Crystal Systems
Crystal systems are categorized based on their axial lengths and interaxial angles. Select a system to explore its geometric properties and interactive 3D lattices.

3 Materials
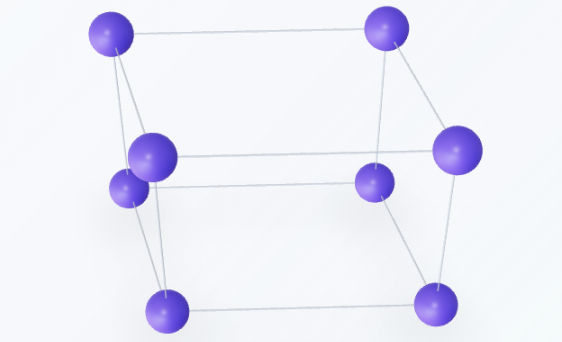
Hexagonal
The hexagonal lattice is defined by a primary axis perpendicular to two base axes of equal length that intersect at a $120^\circ$ angle. This system serves as the foundation for the hexagonal close-packed (HCP) structure, one of the most efficient ways to pack spheres in a crystal.Lattice parameters: $a = b \neq c$, and angles $\alpha = \beta = 90^\circ, \gamma = 120^\circ$.Examples: Magnesium, Zinc, Titanium, and Graphite.
Explore System
0 Materials
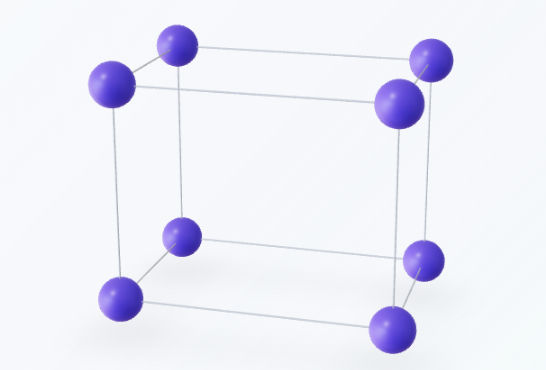
Monoclinic
The monoclinic system is defined by three axes of unequal length. In this geometry, two of the axes intersect at an oblique angle, while the third remains strictly perpendicular to the plane formed by the other two. Because of this specific inclination, the system exhibits relatively low symmetry—typically characterized by a single twofold rotation axis or a single mirror plane—and includes two Bravais lattices: simple monoclinic and base-centered monoclinic.Lattice parameters: $a \neq b \neq c$, and angles $\alpha = \gamma = 90^\circ, \beta \neq 90^\circ$.Examples: Gypsum, Orthoclase, Borax, and Sodium Sulfate.
Explore System
0 Materials
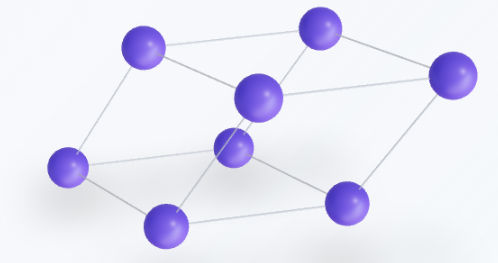
Orthorhombic
The orthorhombic system is defined by three mutually perpendicular axes, all of which have unequal lengths. Geometrically, it takes the shape of a rectangular prism. It is a unique and highly versatile system in crystallography, as it is the only one that accommodates all four Bravais lattice types: primitive ($P$), base-centered ($C$), body-centered ($I$), and face-centered ($F$).From a symmetry perspective, it is characterized by three mutually perpendicular two-fold rotation axes or mirror planes. Because the edge lengths are all different ($a$, $b$, and $c$), physical properties like electrical conductivity or refractive index often vary depending on the direction of measurement, a phenomenon known as anisotropy.Lattice parameters: $a \neq b \neq c$, and angles $\alpha = \beta = \gamma = 90^\circ$.Examples: Alpha-Sulfur (rhombic), Olivine, Topaz, and Aragonite.
Explore System
0 Materials
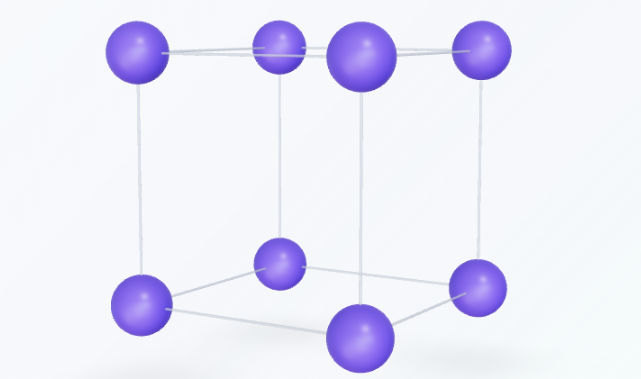
Rhombohedral
Also known as trigonal, the rhombohedral system is defined by three equal axes that are equally inclined to each other at an angle other than $90^\circ$. It can be thought of as a cube stretched along its body diagonal. Lattice parameters: $a = b = c$, and angles $\alpha = \beta = \gamma \neq 90^\circ$. Examples: Quartz, Calcite, Bismuth.
Explore System
9 Materials
Simple Cubic
OverviewThe simple cubic (sc) structure is the simplest crystal lattice system. In this arrangement, atoms are located exactly at the eight corners of a cube. While it is fundamental to understanding crystallography, very few elements (only Polonium, $\text{Po}$) crystallize in this structure under normal conditions because of its low packing efficiency.Geometrical PropertiesThe simple cubic system has equal edge lengths and all angles are right angles. The lattice parameters are defined as: $$ a = b = c $$ $$ \alpha = \beta = \gamma = 90^\circ $$Atomic Packing Factor (APF)The Atomic Packing Factor represents the fraction of volume in a crystal structure that is occupied by constituent particles.For a simple cubic unit cell, the atoms strictly touch along the cube edges. Therefore, the relationship between the atomic radius ($r$) and the lattice constant ($a$) is: $$ a = 2r $$Since there is exactly $1$ atom per unit cell (8 corners $\times \frac{1}{8}$ atom per corner), the total volume of atoms in the cell ($V_{atoms}$) is: $$ V_{atoms} = 1 \times \left( \frac{4}{3} \pi r^3 \right) $$The total volume of the cubic unit cell ($V_{cell}$) is simply: $$ V_{cell} = a^3 = (2r)^3 = 8r^3 $$Thus, the Atomic Packing Factor is calculated as: $$ \text{APF} = \frac{V_{atoms}}{V_{cell}} = \frac{\frac{4}{3} \pi r^3}{8r^3} = \frac{\pi}{6} \approx 0.524 $$ This means that only about $52.4%$ of the unit cell volume is filled with atoms, leaving $47.6%$ as empty space.
Explore System

1 Materials
Tetragonal
The tetragonal crystal system results from stretching a cubic lattice along one of its vectors, so that the cube becomes a rectangular prism with a square base. Lattice parameters: $a = b \neq c$, and angles $\alpha = \beta = \gamma = 90^\circ$. Examples: White Tin, Rutile (TiO$_2$).
Explore System
0 Materials
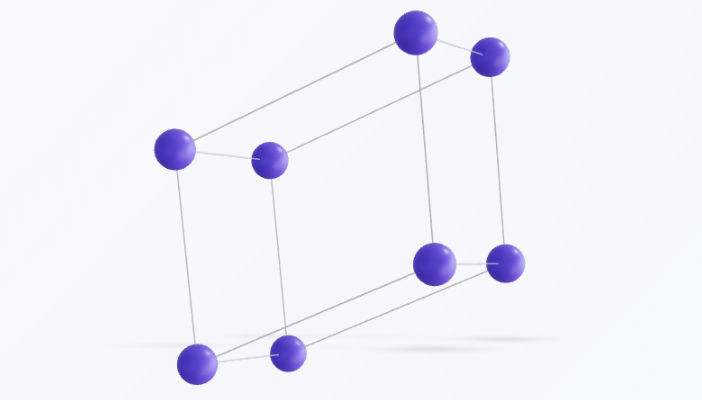
Triclinic
The triclinic system is the least symmetric of the seven crystal systems, characterized by a complete lack of mirror planes or rotational axes. In this geometry, all three crystallographic axes are of unequal length and intersect at oblique angles, meaning no two axes are perpendicular to one another.Key Characteristics & SymmetryFrom a symmetry standpoint, the triclinic system only possesses translational symmetry and, in some cases, inversion symmetry (a center of symmetry). Because of this high degree of asymmetry, it is the only system that allows just a single Bravais lattice: the primitive ($P$) lattice.Physical properties in triclinic crystals—such as thermal expansion or electrical conductivity—are highly anisotropic, meaning they vary significantly depending on the direction in which they are measured.Lattice ParametersTo define a triclinic unit cell, all six parameters must be specified independently:Axial lengths: $a \neq b \neq c$Interaxial angles: $\alpha \neq \beta \neq \gamma \neq 90^\circ$Common ExamplesMicrocline: A common potassium feldspar.Turquoise: A hydrated phosphate of copper and aluminum.Kyanite: An aluminosilicate mineral noted for its distinct blue color.Copper(II) Sulfate Pentahydrate ($CuSO_4 \cdot 5H_2O$): A well-known laboratory chemical.
Explore System